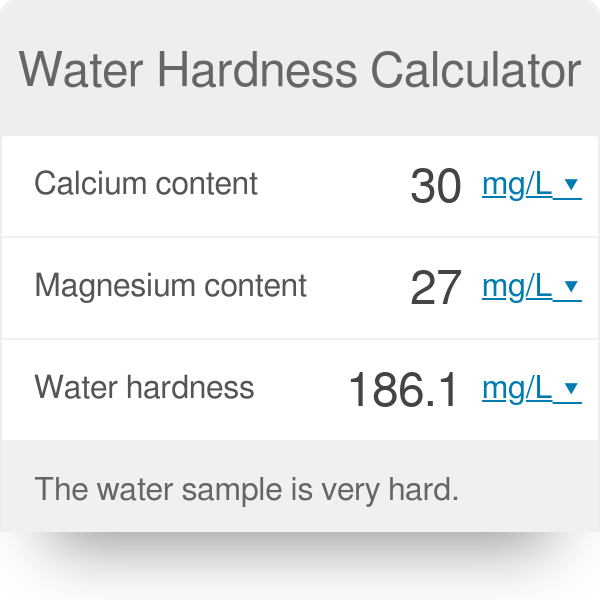
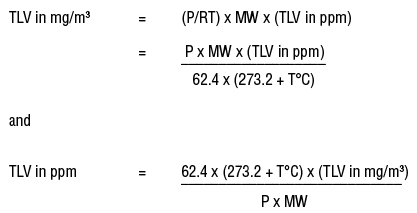
What is the formula to calculate the actual results of a diluted and digested sample by using AAS in PPM | ResearchGate

How to convert arsenic concentration ug/L (in solution) to mg/kg (in soil) after sequential extraction experiment ? | ResearchGate

Pounds Formula Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit. - ppt download

SOLVED: A 190-mg sample of Na2CO3 is dissolved in H2O to give 1.00L of solution. What is the concentration of Na+ in parts per million (ppm)? Note: "ppm" are 106 ×[(mass of
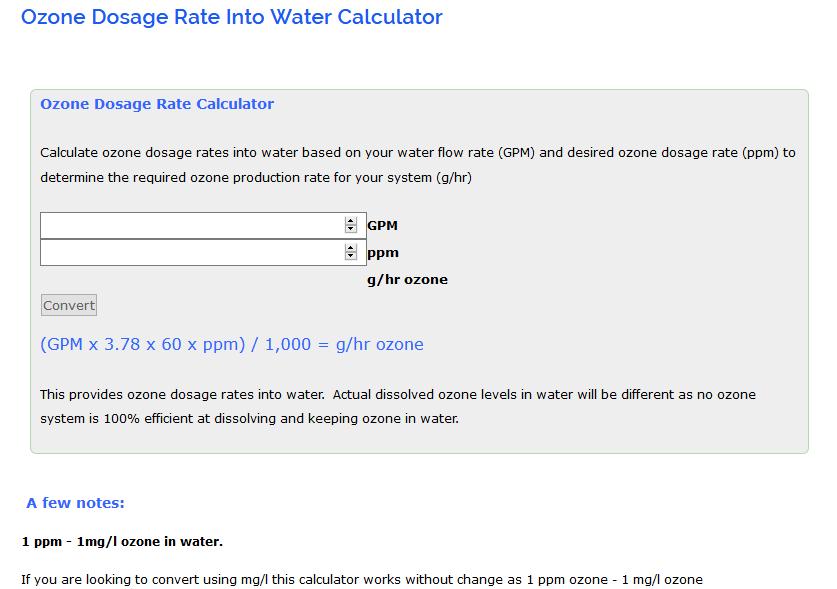
Ozone equipment manufacturer and ozone system integrators Ozone dosage rate into water calculator Ozone Integration Experts



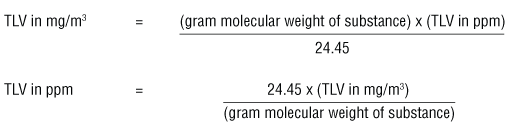
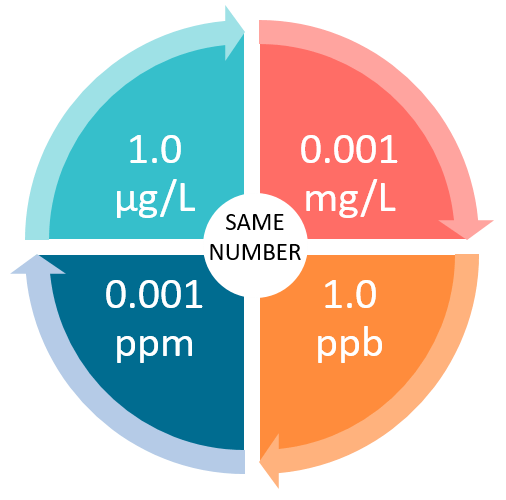


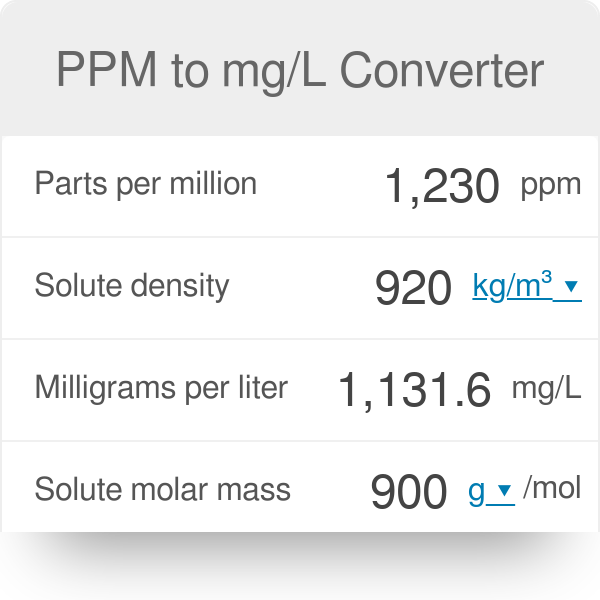

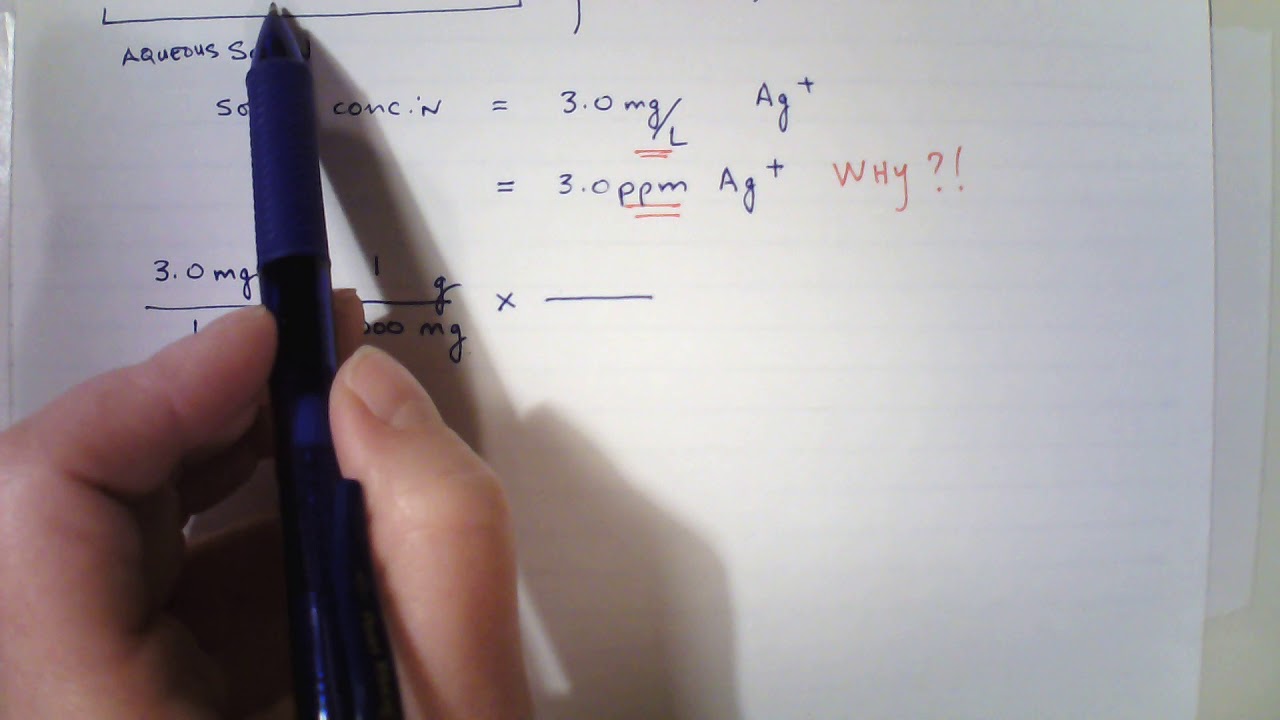





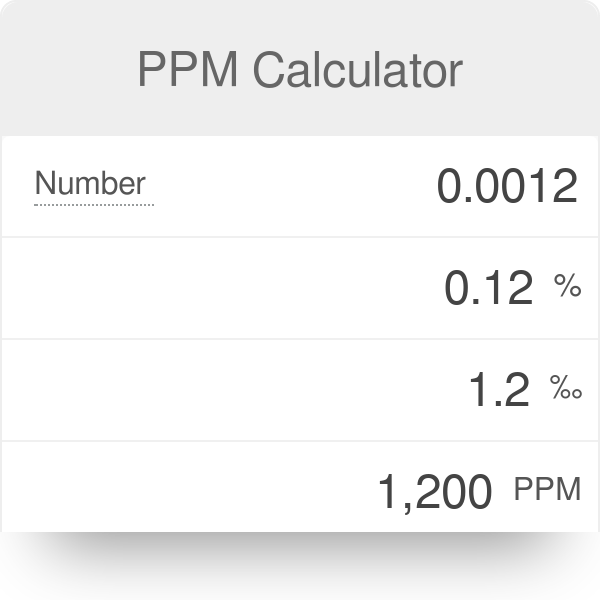
:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)